Mathematical models of cell phenotype regulation and reprogramming: Make cancer cells sensitive again!
Abstract
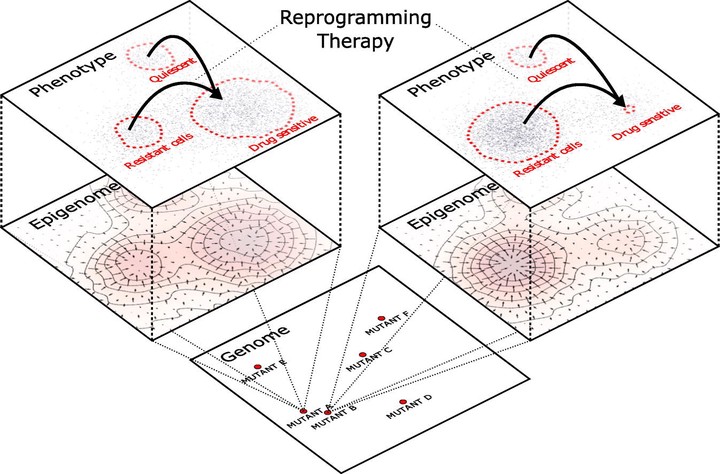
© 2017 A cell’s phenotype is the observable actualization of complex interactions between its genome, epigenome, and local environment. While traditional views in cancer have held that cellular and tumor phenotypes are largely functions of genomic instability, increasing attention has recently been given to epigenetic and microenvironmental influences. Such non-genetic factors allow cancer cells to experience intrinsic diversity and plasticity, and at the tumor level can result in phenotypic heterogeneity and treatment evasion. In 2006, Takahashi and Yamanaka exploited the epigenome’s plasticity by “reprogramming” differentiated cells into a pluripotent state by inducing expression of a cocktail of four transcription factors. Recent advances in cancer biology have shown not only that cellular reprogramming is possible for malignant cells, but it may provide a foundation for future therapies. Nevertheless, cell reprogramming experiments are frequently plagued by low efficiency, activation of aberrant transcriptional programs, instability, and often rely on expertise gathered from systems which may not translate directly to cancer. Here, we review a theoretical framework tracing back to Waddington’s epigenetic landscape which may be used to derive quantitative and qualitative understanding of cellular reprogramming. Implications for tumor heterogeneity, evolution and adaptation are discussed in the context of designing new treatments to re-sensitize recalcitrant tumors. This article is part of a Special Issue entitled: Evolutionary principles — heterogeneity in cancer?, edited by Dr. Robert A. Gatenby.